EPDM - The all-round rubber: High resistance to weathering, ageing and ozone
1 What is EPDM?
EPDM (ethylene propylene diene monomer rubber) is a synthetic, largely saturated elastomer based on a terpolymer of ethylene, propylene and a diene monomer that enables sulphur vulcanisation.
EPDM is characterised by very high resistance to ozone, UV radiation, weathering, hot water and many polar media. The typical application temperature range is between around -50 °C and +130 °C, and even higher for special compounds.
Due to its saturated polymer structure, EPDM boasts excellent ageing stability, but has limited resistance to mineral oils, fuels, and aromatic hydrocarbons. Typical applications include seals, profiles, Buffer of rubber and RUBBER AND METAL) Membranes, hoses, and external applications in construction, the automotive industry, and plant engineering.
How was EPDM developed?
The development of EPDM rubber dates back to the late 1950s and early 1960s and is closely linked to the progress of Ziegler-Natta catalyst technology. This was the first to enable the controlled copolymerisation of ethylene and propylene and the targeted incorporation of a diene monomer into the polymer structure.
EPDM was the first material to combine the high environmental resistance of saturated polyolefins with the elasticity and cross-linkability of classic rubbers. This meant that the limited ozone and ageing resistance of many previously used elastomers could be significantly improved.
Advances in polymerisation, compound design and cross-linking technology have steadily expanded the range of available EPDM grades and turned the material into a versatile elastomer system.
Chemical structure: What is the chemical structure of EPDM?
EPDM rubber is a terpolymer made from ethylene (E), propylene (P) and an unsaturated diene monomer (DM). The dienes used are mainly ethylidene norbornene (ENB), dicyclopentadiene (DCPD) or 1,4-hexadiene. The choice of diene monomer particularly influences the cross-linking ability and the reaction behaviour during vulcanisation.
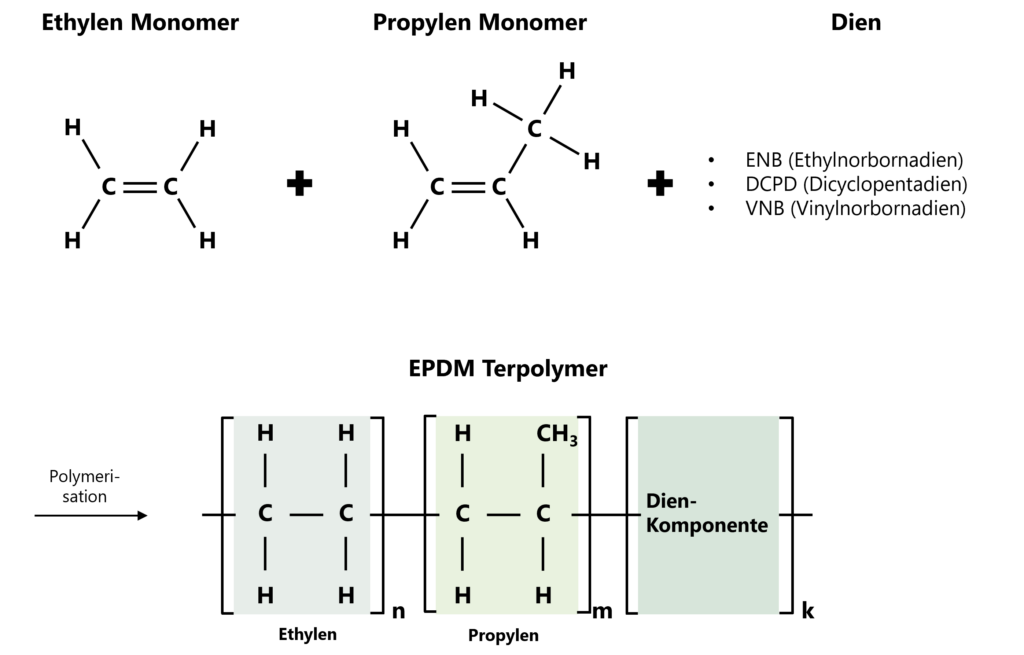
Figure 1: Polymer structure of the ethylene-propylene-diene monomer (EPDM) as a terpolymer consisting of an ethylene monomer (top left), a propylene monomer (top centre) and a diene (top right).
The EPDM main chain consists entirely of saturated carbon-carbon bonds and is largely responsible for the high chemical stability as well as the pronounced resistance to ozone, UV radiation and oxidative ageing. The double bonds required for cross-linking are introduced into the side chains exclusively via the diene portion.
The ratio of ethylene to propylene determines the properties of the polymer. Increasing ethylene content promotes the formation of crystalline areas and increases hardness and tensile strength, while higher propylene content improves elasticity and low-temperature flexibility. In combination with the diene monomers, a terpolymer is formed whose physical properties can be adjusted by the composition without impairing the chemical stability of the saturated main chain.
What are the differences between EPDM with low and high diene content?
The diene content determines the number of cross-linkable double bonds and influences the vulcanisation behaviour. A higher diene content enables stronger cross-linking between the polymer chains and higher Shore strengths, but at the same time leads to lower resistance to ageing and weathering. In practice, the diene content of commercially available EPDM rubber is in the low single-digit to low double-digit percentage range by weight.
How is EPDM cross-linked?
Cross-linking is a key step in the processing of EPDM compounds, as it enables the plastic rubber compound to be converted into an elastic, permanently dimensionally stable EPDM rubber
During sulphur vulcanisation, flexible sulphur bridges are formed between the polymer chains. This network structure gives the resulting EPDM rubber high elasticity and good dynamic properties, especially under changing loads. Sulphur-cured EPDM is characterised by good resilience, but shows an increased tendency to compression set at elevated temperatures and under long-term mechanical stress.
Peroxide cross-linking leads to carbon-carbon cross-links. These are shorter and more thermally stable than sulphur bridges and lead to a more densely cross-linked, chemically resistant network. Peroxide cross-linked EPDM has better heat ageing resistance, lower permanent deformation and higher long-term stability under thermal stress.
The choice of crosslinking system is therefore less a question of the base polymer than of the desired combination of properties. It is determined early on in the compounding process and influences both the processing and the subsequent properties of the EPDM component.
How does the ethylene content influence the material properties?
- Low ethylene content (approx. 45-55 wt.-%): predominantly amorphous structure with high elasticity and very good low-temperature flexibility.
- Medium ethylene content (approx. 55-65 wt.-%): semi-crystalline structure with a good balance between strength, elasticity and workability.
- High ethylene content (> 65 wt.-%): pronounced crystalline areas with increased tensile strength even in the non-crosslinked state.
2. chemical and thermal EPDM properties
EPDM is characterised by a distinctive combination of mechanical performance, high environmental resistance and chemical stability, especially under permanent exposure to weather and the environment.
The key material properties of EPDM include
| Superior chemical and thermal resistance | Medium resistance | Limited durability |
|---|---|---|
| Weather conditions | Hot water | Mineral oil |
| Ozone | Water vapour | Fat |
| UV | Acids | Petrol / Fuels |
| Temperature | Lye | Aromatic hydrocarbons |
| Polar media |
Table 1: Primary material properties of EPDM
EPDM has good electrical insulating properties and is therefore also suitable for use in electrotechnical applications, such as cable sheathing, seals or insulating components where reliable separation of conductive components is required.
EPDM can also be adjusted over a wide hardness range via the compounding composition. Typical EPDM compounds cover a range from around 20 to 95 Shore A and therefore enable both very soft-elastic and significantly more dimensionally stable designs for different technical requirements.
Mechanically, EPDM achieves high elongation at break of up to 500 % and tensile strengths in the medium range, depending on the compound and cross-linking. Due to its low density, EPDM is one of the lighter elastomer materials.
A key feature is the long-term stability of elasticity, even under changing environmental conditions and at low temperatures. At the same time, EPDM, especially in sulphur-cured versions, has a higher tendency to compression set under continuous load. This effect can be specifically reduced by using a suitable formulation and peroxide crosslinking.
For applications with very high gas-tightness requirements, this can be a limiting factor, whereas in many other applications it is not a practical restriction.
Shore hardening for EPDM
The hardness of the EPDM material is usually measured according to Shore A. The adjustable hardness range is typically between 20 Shore A and 95 Shore A and can be adjusted by formulation, filler system, type of cross-linking and degree of cross-linking. EPDM 70, for example, refers to a combination of the material EPDM with a nominal hardness of 70 Shore A.
EPDM in the range of around 20 to 40 Shore A is used for applications with high flexibility requirements, for example for sealing lips, membranes or vibration elements. EPDM material of this Shore hardness has high mouldability and good low-temperature flexibility, but has lower mechanical rigidity and higher permanent deformation under load.
Hardness ranges of around 50 to 70 Shore A are standard for sealing and profile applications, such as O-rings, flat gaskets, window and façade profiles and moulded parts. In this range, there is a balanced relationship between elasticity, strength, abrasion behaviour and compression set.
Highly hard EPDM materials, in the range of around 80 to 95 Shore A, are used when high dimensional stability and low elastic deformation under mechanical load are required, for example in support rings, wipers, guide and bearing components.
The required Shore hardness is determined based on the application, taking into account the mechanical load, installation situation, temperature range, media contact and service life requirements. In practice, the target hardness is defined as the starting parameter for the compound design and then verified by component tests.
Standards & guidelines for EPDM
EPDM is not a standardised material, but is clearly classified according to international classification, testing and evaluation standards. The basic definition is based on ISO 1629 and ASTM D1418, in which EPDM rubber is described as ethylene-propylene-diene rubber and is assigned to group M (saturated polymethylene main chain).
In practice, the technical properties of EPDM materials are mainly specified on the basis of general elastomer test standards. These include standards for tensile strength, hardness, tear propagation resistance and compression set (e.g. ISO 37, ISO 48, ISO 34, ISO 815).
Another focus is on ageing and environmental resistance. Tests in accordance with ISO 188 (heat ageing) and ISO 1431-1 (ozone resistance) are not specific to EPDM, but address the loads for which EPDM is typically designed.
For specific components and application contexts, application-specific regulations also come into play. Standards such as DIN ISO 3601 (O-rings) or DIN 7863 (elastomer profiles) define geometric and qualitative requirements without specifying the material. For sensitive areas of application, such as contact with drinking water or foodstuffs, assessment is also carried out using regulations such as KTW-BWGL or WRAS, according to which EPDM compounds can be tested and approved.
Accordingly, there is no universally valid „EPDM standard“, but rather a standard-compliant design of the EPDM compound and subsequent testing and approval of the resulting EPDM material with regard to the intended conditions of use.
Purity and cleanliness requirements for EPDM
In addition to mechanical and chemical characteristics, EPDM compounds can also be designed with regard to different levels of purity and cleanliness. These are not regulated by separate EPDM standards, but result from the composition of the compound, the selection of raw materials and the process control during mixing, processing and post-treatment, focussing in particular on how many migratable components, low-molecular residues and particulate impurities remain in the material after processing.
Compliance with defined purity requirements for EPDM therefore depends more on the compound design and production. It requires controlled raw materials, reproducible mixing processes and clearly defined quality criteria along the entire process chain.
3. processing & production of EPDM
EPDM materials can be reliably processed using all standard elastomer processing methods and are particularly suitable for industrial series production. The choice of manufacturing process depends primarily on the component geometry, quantity and the required material properties.
For moulded parts Compression presses, Transfer presses and Injection moulding process for use. All three variants enable high dimensional accuracy, reproducible component properties and economical production, even with complex geometries. Profiles and hoses are usually extruded, often with downstream continuous vulcanisation.
EPDM is characterised by stable flow behaviour, uniform cross-linking and comparatively low shrinkage. These properties make the material process-safe, but require a precisely coordinated formulation and controlled vulcanisation parameters.
4. material comparison: EPDM vs. other elastomers
Compared to other common elastomers, EPDM is resistant to environmental influences and ageing.
| International abbreviation | EPDM | NBR | CR | FKM | |
| Hardness range (in Shore) | 20A – 95A | 20A – 75D | 20A – 90A | 50A – 90A | |
|
Mechanical Properties for Room temperature |
Tear resistance | 3 | 3 | 3 | 2 |
| Elongation at break | 3 | 3 | 3 | 2 | |
| Rebound resilience | 3 | 2 | 2 | 0 | |
| Tear propagation resistance | 3 | 2 | 3 | 1 | |
| Abrasion resistance | 3 | 2 | 3 | 2 | |
|
Pressure deformation residue |
with max. continuous use temperature |
0 | 1 | 2 | 0 |
| at room temperature | 0 | 0 | 0 | 1 | |
| Thermal behaviour | Cooling behaviour (Tg) up to °C | -50 | -30 | -40 | -30 |
| Max. Continuous operating temperature up to °C | 130 | 110 | 100 | 220 | |
| Resistance to | Petrol | 1 | 2 | 2 | 3 |
| Mineral oil (at 100 °C) | 1 | 3 | 2 | 3 | |
| Acids (aqueous inorganic acids at RT) | 3 | 2 | 2 | 3 | |
| Alkalis (aqueous inorganic alkalis at RT) | 3 | 2 | 2 | 3 | |
| Water (at 100 °C, distilled) | 3 | 2 | 2 | 3 | |
| Weather and ozone | 3 | 2 | 2 | 3 | |
Table 1: Material comparison of EPDM with NBR, CR and FKM
What does EPDM rubber cost?
Together with NBR, EPDM is one of the comparatively inexpensive rubber raw materials. The purchase prices for the raw material can vary greatly depending on the compounding, the quantity purchased, the additives and the certifications for specific applications.
| Material | EPDM | NBR | CR | TPE/TPU | Silicone (LSR) | Silicone (HTV) | HNBR | FKM |
|---|---|---|---|---|---|---|---|---|
| Cost factor | x 1,0 | x 1,0 | x 1,2 | x 1,3 | x 1,4 | x 1,8 | x 2,9 | x 3,7 |
Differences between EPDM and NBR
Compared to acrylonitrile butadiene rubber (NBR), ethylene propylene diene rubber (EPDM) offers significantly better resistance to weathering, ozone and UV radiation. NBR, on the other hand, is designed for contact with media containing oil and fuel, while EPDM is unsuitable in such environments due to strong swelling.
Differences between EPDM and CR
Compared to chloroprene rubber (CR), EPDM is characterised by greater long-term stability and better low-temperature properties. CR, on the other hand, offers advantages in terms of moderate oil resistance and flame-retardant behaviour, but does not achieve the same resistance to permanent weathering.
Differences between EPDM and FKM
Compared to fluororubber (FKM), EPDM is significantly more cost-efficient, but has clear disadvantages in terms of temperature and media resistance. FKM is resistant to a wide range of aggressive media and can also be used at higher temperatures. EPDM, on the other hand, is used where environmental resistance is paramount and no highly reactive media are present.
Mehr zum Material EPDM und zur Produktion von Gummiform- und Verbundteilen finden Sie in unserem ausführlichen Gummi-Lexikon, dem Glossary.
