SBR (styrene-butadiene rubber) - solid mechanical properties for universal moulded rubber parts
1. introduction & history
SBR (styrene-butadiene rubber) is a synthetic copolymer rubber made from styrene and butadiene, which is produced by emulsion or solution polymerisation and belongs to the group of unsaturated synthetic rubbers.
Typical characteristics of the material are good abrasion resistance, high elasticity, good ageing resistance and a good balance between mechanical strength and cost-effectiveness. The temperature resistance is generally between -45 °C and +90 °C.
SBR is mainly used for tyre treads, technical moulded rubber parts, seals and vibration dampers. Compared to natural rubber (NR), SBR offers better heat and ageing resistance, but does not achieve its tear resistance and dynamic load capacity. Compared to oil-resistant rubbers such as NBR, SBR is only resistant to mineral oils and fuels to a limited extent.
The history of SBR is closely linked to the shortage of raw materials during the Second World War. When access to natural rubber from tropical regions was restricted, Germany, the USA and later other industrialised nations in particular intensified their research into synthetic alternatives. SBR quickly became a strategically important substance that met the demand for wear-resistant rubber material.
Butadiene and styrene, the raw materials for SBR, come from the cracking of crude oil or natural gas, which means that their price and availability are closely linked to developments in fossil raw materials. Despite this dependency, SBR remains one of the most important materials in rubber processing due to its high availability, good processing properties and stable material quality.
2. chemical composition of SBR
The styrene-butadiene copolymer consists of the two monomers styrene (C₈H₈) and 1,3-butadiene (C₄H₆).
Styrene is an unsaturated aromatic hydrocarbon consisting of a benzene ring with a vinyl group. In the chemical industry, styrene is present as a reactive, colourless and oily liquid with a characteristic sweet odour. Its physical properties include a density of approx. 0.91 g/cm³, a boiling point of around 145 °C and good solubility in organic solvents, but low solubility in water.
Butadiene is a conjugated diene hydrocarbon with two double bonds, which is present as a colourless gas and has a slightly sweet smell. It has a density of 0.62 g/cm³ (at 0 °C), a boiling point of approx. -4.4 °C and is easily liquefied under pressure.
These two monomers are (co-)polymerised with each other in variable proportions, typically in a weight ratio of around 23% styrene to 77 % butadiene. The exact composition largely determines the material properties: a higher proportion of styrene increases the hardness and abrasion resistance, while a lower proportion improves the elasticity and low-temperature flexibility.
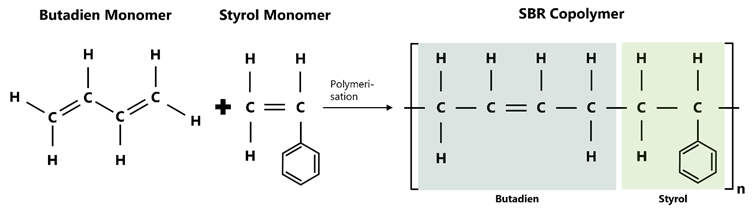
Figure 1: Polymer structure of styrene-butadiene rubber (SBR) as a copolymer of butadiene (left) and styrene units (right), which are repeatedly arranged in the chain.
At the molecular level, SBR consists of randomly arranged copolymer chains in which the styrene and butadiene units are randomly distributed in the polymer chain. The arrangement of the butadiene units can occur in 1,4- (cis/trans) and 1,2-vinyl form, whereby 1,4-trans proportions with 60-70% predominate. The 1,2-vinyl portion with the double bond in the side chain plays a particularly important role, as it significantly influences the glass transition point (Tg) and thus the temperature behaviour of the material.
Polymerisation process for styrene-butadiene rubber
Styrene-butadiene rubber is mainly produced by two types of polymerisation:
- Emulsion polymerisation (E-SBR) is carried out in an aqueous phase using emulsifiers and initiators (e.g. peroxides) - a proven, cost-effective process for standard grades. The structure of the copolymer is formed relatively randomly with this method and fine control is not possible.
- Solution polymerisation (S-SBR), in which organic solvents and lithium catalysts are used to specifically control the microstructure (styrene content, cis/trans/vinyl content). This variant delivers products with narrower property tolerances and therefore also higher performance.
Physical appearance of styrene-butadiene rubber
The raw material is usually in the form of grainy, porous granules in a white to light yellow/brownish colour. The surface is slightly sticky, which is due to the residual oil or plasticiser content that keeps the rubber workable.
Vulcanisation mechanisms of styrene-butadiene rubber
Various vulcanisation variants are used to crosslink SBR, depending on the application requirements. The most common is sulphur vulcanisation, in which sulphur in combination with accelerators and activators at temperatures between 140 and 180 °C forms covalent bridges of up to eight atoms in length between the polymer chains. Sulphur vulcanisation offers a balanced combination of abrasion resistance, elasticity and economy and is therefore the standard both in the tyre industry and for technical rubber goods.
From a chemical-technical point of view, styrene-butadiene rubber is unsaturated, which means that double bonds remain in the butadiene segments. These are essential for vulcanisation, which gives SBR its final strength and elasticity. At the same time, these remaining double bonds make the material more sensitive to ozone and UV exposure unless suitable anti-ageing agents are used.
Peroxide vulcanisation is often chosen for applications with higher thermal loads. Here, direct cross-links are created between the carbon atoms of the polymer chains, which are significantly more resistant to heat and ageing than sulphur bridges. Peroxide vulcanisation is particularly suitable for seals or components that are operated in hot environments, although it is often less optimal for dynamically highly stressed parts.
Another option is resin vulcanisation with reactive phenolic resins, which is used in combination with halogen activators. This process produces very heat-resistant and dimensionally stable rubber parts with low compression set and is preferred for conveyor belts, roller coverings or engine compartment components.
Radiation crosslinking is also used for special applications, such as in medical technology or food contact. In this process, the finished moulded SBR component is treated with electron or gamma rays to create a uniform and chemical-free cross-linking network. While the molecules of the resin provide the cross-linking bridges in resin cross-linking, direct C-C bonds are created between the polymer chains in radiation cross-linking. This method enables very clean end products, but is only established in niche markets due to the system technology and the limited penetration depth.
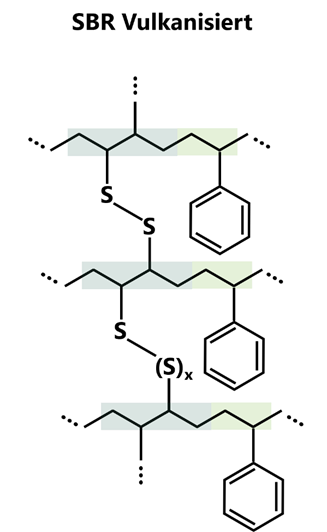
Figure 2: Schematic representation of the possible arrangements of butadiene (left) and styrene units (right) in styrene-butadiene rubber (SBR) and their cross-linking by sulphur bridges.
4. properties & special features of styrene-butadiene rubber
SBR is often used as an alternative to other common rubber types such as natural rubber (NR), acrylonitrile butadiene rubber (NBR) or ethylene propylene diene monomer (EPDM). The choice of material depends heavily on the required properties, the conditions of use and the costs.
| International abbreviation | SBR | NR | EPDM | NBR | |
| Hardness range (in Shore) | 20A-95A | 25A-70D | 20A-95A | 20A-75D | |
| Mechanical properties for room temp. |
Tear Strength |
6 | 8 | 5 | 6 |
| Tear elongation |
6 | 8 | 5 | 6 | |
| Rebound Elasticity |
6 | 8 | 6 | 4 | |
| Tear-away resistance |
4 | 8 | 4 | 4 | |
| Abrasion resistance |
6 | 5 | 5 | 7 | |
| Print deformation rest |
with max. continuous use Temperature |
4 | 4 | 2 | 3 |
| for room temperature |
3 | 2 | 0 | 2 | |
| Thermal Behaviour |
Refrigeration behaviour (Tg) to °C |
-45 | -55 | -50 | -45 |
| Max. Continuous operational Temperature up to °C |
90 | 80 | 130 | 110 | |
| Resistance to | Petrol | 0 | 0 | 0 | 5 |
| Mineral oil (at 100 °C) | 0 | 0 | 0 | 8 | |
| Acids (aqueous inorganic acids at RT) | 2 | 2 | 8 | 3 | |
| Alkalis (aqueous inorganic alkalis at RT) | 3 | 4 | 8 | 2 | |
| Water (at 100 °C, distilled) | 3 | 3 | 8 | 4 | |
| Weather conditions and Ozone |
3 | 3 | 8 | 2 | |
Table 1: Properties of selected materials: 0 = unsuitable, 8 = very suitable
SBR is an all-rounder among synthetic rubbers and combines high abrasion resistance, good low-temperature flexibility, stable mechanical properties and broad formulation flexibility. It is less dynamically resistant than natural rubber, less oil-resistant than NBR and less weather-resistant than EPDM, but surpasses them in abrasion resistance and low-temperature elasticity. Due to its comparatively low cost, SBR offers a good price-performance ratio.
5 Areas of application for styrene-butadiene rubber
Styrene-butadiene rubber (SBR) is firmly established in numerous key industries thanks to its abrasion resistance, elasticity and economical processability.
- Tyre industry
Around 50-60 % of global SBR production is used for car and truck tyres. Emulsion-polymerised SBR is mainly used in standard tyres, while solution-polymerised SBR is essential for premium tyres with low rolling resistance and improved wet braking performance. These properties are crucial for meeting the strict EU tyre label requirements. - Technical rubber goods
SBR is the basis for numerous industrial moulded and extruded parts. These include seals, hoses, profiles, conveyor belts, rubber rollers and roller coverings. The high abrasion and dimensional stability ensures a long service life and low maintenance costs - a particular advantage in demanding production and conveying processes. - Construction industry
In the construction industry, SBR is mainly used for seals, joint profiles and bridge bearings. Its elasticity, even at low temperatures, makes it ideal for outdoor use in Central European climate zones. It is also resistant to weathering if appropriate anti-ageing agents are included in the formulation. - Consumer goods and sports applications
SBR is used in shoe soles and sports floors. Here, good adhesion, wear resistance and flexibility are among the desired properties. By combining them with fillers and plasticisers, the products can be optimally adapted to their respective application.
