IIR rubber: High gas impermeability, good damping and strong weather resistance
1. What is Butyl rubber (IIR)?
Butyl rubber, chemically „isobutene-isoprene rubber“, or IIR for short, is a synthetic elastomer known for its very low gas permeability and its resistance to oxygen, ozone, and many other chemicals. IIR is a copolymer comprising isobutene monomers and a small proportion of isoprene monomers. Due to its long-term stability, gas-tightness, and resistance to chemicals, it is often used in sealing and membrane applications.
History of Butyl Rubber (IIR)
In the 1930s, IIR was developed by researchers as an alternative to natural rubber (NR). In the 1940s, IIR rubber was finally produced industrially and primarily used for tyre inner tubes, as IIR has significantly lower gas permeability compared to NR. In the following decades, IIR was further developed in various forms and variations, expanding its properties for many application areas. Therefore, IIR is still one of the most important synthetic rubbers worldwide today and is used due to its high gas impermeability and its long-term resistance in demanding application areas such as seals in the construction and automotive industries, medical technology, or in chemical apparatus.
Chemical composition of butyl rubber (IIR)
Butyl rubber is a so-called copolymer, consisting of the monomers isobutene and isoprene, where isobutene makes up approximately 95–99 mol%, while isoprene is present in a significantly lower molarity at about 1–5 mol%. The isobutene portion forms the long-chain, completely saturated polymer chain, while the isoprene units represent sites of attack for later vulcanisation due to the double bonds they contain. Butyl rubber has a chemically special structure, thereby creating a very dense polymer responsible for its high gas impermeability and ageing resistance.
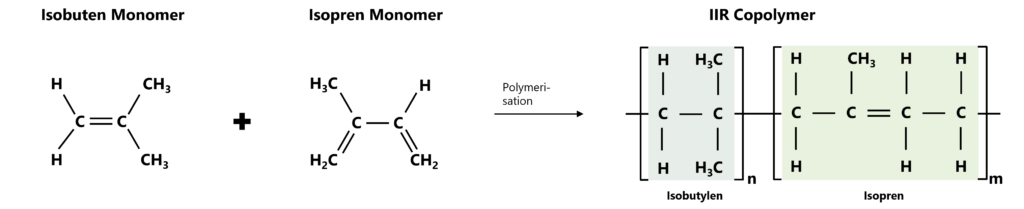
Figure 1: Chemical structure of IIR: From an isobutene monomer (left) and an isoprene monomer (centre) The IIR copolymer (right) is formed by polymerisation.
2. Properties and characteristics of butyl rubber (IIR)
The properties of IIR can be divided into the categories of „chemical properties“, „mechanical properties“, and „physical properties“. Furthermore, modified butyl rubbers exist which are used especially in demanding applications.
Chemical properties of IIR
Butyl rubber is chemically very resistant to oxygen, ozone, and many other chemicals, particularly acids, bases, and oxidising agents. However, butyl rubber is sensitive to aromatic hydrocarbons, mineral oils, and fats. Upon contact with these substances, the material begins to swell or exhibits mechanical weaknesses. Due to the low density of double bonds in the polymer, IIR is very stable against ageing and ozone, but this also makes vulcanisation more difficult, as double bonds must be broken and then new bonds formed in other directions. With a low density of double bonds, the degree of cross-linking is lower, and the mechanical properties are impaired.
While IIR is not considered a classic „biocompatible“ elastomer, butyl rubber is used in biomedical applications depending on the type, residual monomers, additives, and the nature of contact (skin, blood, implant, etc.) after system-specific testing according to the ISO 10993 standard. In practice, IIR can be particularly useful for short-term or indirect contact, for example, as stoppers or in closures.
For applications in contact with foodstuffs, special IIR compounds must be chosen that meet the relevant requirements such as EU-1935/2004, FDA listings, and positive lists for additives. For drinking water applications, additional national regulations such as KTW-BWGL, DVGW worksheets, and VDI 6023 (in the case of Germany) must be complied with. In principle, IIR components are therefore possible in drinking water and food applications, but not every IIR material is automatically approved for them.
Mechanical properties of IIR
Mechanically, IIR exhibits high elasticity, good tensile strength, and good elongation. Typical IIR components show elongations at break in the range of approximately 400 to 800% of the original size, sometimes even higher. The hardness of IIR ranges from 30 to 80 Shore A, depending on the formulation, enabling flexible yet dimensionally stable products. At the same time, this softness of IIR is also reflected in lower abrasion resistance compared to natural rubber. Furthermore, IIR shows limited dynamic load-bearing capacity at very high frequencies, which can have a detrimental effect in heavily stressed tyre tread or damping elements.
Physical properties of IIR
The exceptionally low gas permeability is a key characteristic of butyl rubber. IIR exhibits a gas permeability approximately ten times lower than natural rubber, making the material ideal for butyl rubber seals, inner tubes, or gas-tight membranes. Furthermore, IIR possesses good electrical insulation properties and a wide working temperature range from -60 °C to +130 °C. Within these temperatures, the mechanical properties remain largely intact, and the performance of the butyl rubber component is maintained. A disadvantage, in particular, is its low thermal conductivity. This can lead to problems, especially in applications with thermal stress, as it can cause local heat hotspots.
IIR is electrically insulating without fillers and special additives and is therefore frequently used for cable insulation and electrical seals. By adding suitable fillers such as carbon black, graphite, or carbon black/fibre combinations, antistatic to conductive materials can be formulated. Depending on the mixture, the electrical characteristics of the material can thus be individually adapted.
Modified butyl rubbers
Modified variants of IIR are primarily halogenated butyl rubber types such as bromobutyl (BIIR) and chlorobutyl (CIIR), which are produced by halogenation of standard IIR in solution. Bromine or chlorine atoms are incorporated into the isoprene units, resulting in a significantly altered activity profile. BIIR exhibits considerably faster vulcanisation, leading to higher crosslink densities, better resilience properties, and shorter cycle times. CIIR combines the properties of BIIR with additionally very good resistance to thermal ageing, low permanent set, and excellent adhesion in tyre inner liners and carcass compounds.
Mechanically, BIIR/CIIR variants exhibit improved tensile strength, lower permanent deformation, and better fatigue resistance, coupled with good airtightness. In dynamic applications, components made from halogenated butyl rubbers are particularly attractive for highly dynamic areas of use such as tyres, vibration dampers, and membranes due to their finer molecular networks.
3. Processing of Butyl Rubber (IIR)
Polymerisation
IIR is produced by cationic polymerisation of isobutene with the aid of a catalyst such as aluminium(III) chloride or boron trifluoride. The catalyst activates the monomer, which then forms the polymer chain. To achieve precisely controllable and consistently uniform properties and qualities of the material, butyl rubber is polymerised at low temperatures of approximately -90 to -100 °C. The low temperatures enable a very regular, linear polymer structure that contributes to very high gas impermeability in applications such as butyl rubber seals. The isoprene content in the polymer is added in a controlled manner to ensure the desired double bond density and thus the vulcanisation capability.
Vulcanisation process of IIR
The vulcanisation of IIR rubber is usually carried out using sulphur systems. The relatively low proportion of double bonds leads to relatively slow cross-linking reactions. At temperatures of approximately 120 to 160 °C, the double bonds in the polymer are broken by sulphur atoms, and new, cross-linking sulphur bridge bonds are formed between the polymer chains. This structural change creates a mechanically and thermally more stable network.
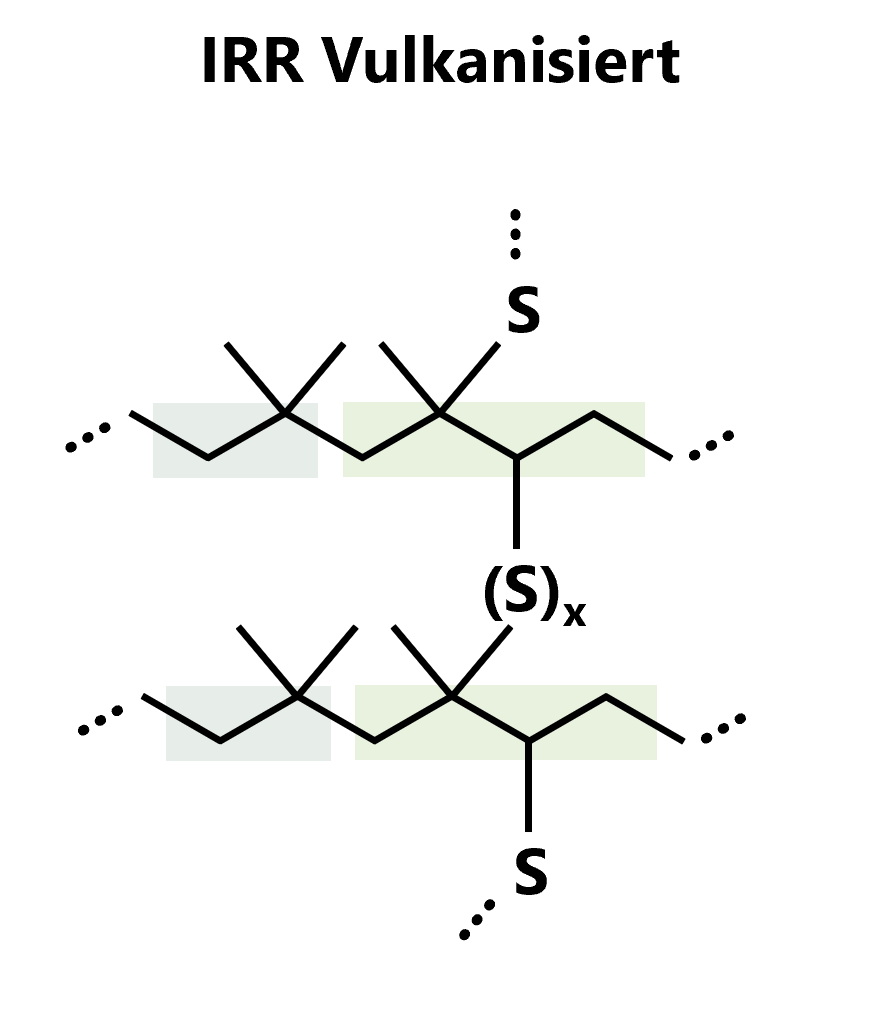
Figure 2: Vulkanised form of IIR
Modified, halogenated variants such as chlorobutyl rubber (CIIR) or bromobutyl rubber (BIIR) can be used for faster and more uniform crosslinking because the halogen atoms increase reactivity. However, disadvantages include, for example, increased susceptibility to gas entrapment due to the higher gas tightness of the IIR material. To avoid these defects, compositions are carefully tuned to achieve optimal network density.
Technical processing of IIR
Technically, IIR is mainly processed using mixing and vulcanisation equipment. This allows for the production of butyl rubber with fillers, plasticisers, and vulcanising agents. Due to its relatively high viscosity and good gas impermeability, IIR can be processed particularly well using extrusion, calendering, and injection moulding. However, this often requires longer mixing times and careful venting to avoid gas inclusions. Furthermore, IIR is frequently mixed with other elastomers such as NR or EPDM to optimise processability, dynamics, and cost. This allows for the production of excellent butyl rubber seals with high long-term stability, gas impermeability, and high resistance to chemicals.
4. Material comparison between butyl rubber (IIR) and other elastomers
If one compares the butyl rubber properties with alternative rubbers, IIR is characterised above all by the highest gas tightness. In general, IIR shows very high resistance to aqueous inorganic acids and alkalis at room temperature, but IIR remains behind FVMQ, VMQ, FKM, HNBR, CR and EPDM in terms of resistance to weathering and ozone. Under mechanical stress, a distinction must be made between very good elongation at break behaviour (similar to VMQ or NR) and comparatively weak abrasion resistance (HNBR > NBR > CR > SBR > EPDM > NR > FKM > IIR > FVMQ > VMQ). In terms of hardness, IIR variants can be formulated from 30 Shore A to 80 Shore A. This puts IIR rubber in the middle of the adaptation range, but lower than other rubbers (e.g. NR, CR, VMQ, SBR, EPDM, NBR), which can be adapted more widely in terms of hardness. Thermally, butyl rubber is particularly characterised by its ability to maintain its properties down to -60 °C. This performance is maintained up to around 130 °C. IIR can therefore be used in a wide range of highly stressed applications - for example as tyre inner tubes (in the case of strong temperature fluctuations), seals, membranes, tank linings and protective clothing (e.g. gloves), in the pharmaceutical industry (closures, plugs), electrical industry (cable insulation), automotive industry (vibration dampers) or in sporting goods (balls).
| International abbreviation | IIR | IR | EPDM | SBR | |
| Hardness range (in Shore) | 30A–80A | 30A–95A | 20A-95A | 20A-70D | |
| Mechanical properties at room temp. | Tear resistance | 2 | 3 | 3 | 3 |
| Elongation at break | 4 | 3 | 3 | 3 | |
| Rebound resilience | 0 | 4 | 3 | 3 | |
| Tear propagation resistance | 3 | 3 | 3 | 3 | |
| Abrasion resistance | 3 | 2 | 1 | 1 | |
| Compression set | at max. continuous operating temperature | 4 | 1 | 0 | 1 |
| at room temperature | 2 | 0 | 0 | 0 | |
| Thermal behaviour | Cooling behaviour (Tg) up to °C | -60 | -55 | -50 | -45 |
| Max. Continuous operating temperature up to °C | 130 | 80 | 130 | 90 | |
| Resistance to | Petrol | 1 | 1 | 1 | 1 |
| Mineral oil (at 100 °C) | 1 | 1 | 1 | 1 | |
| Acids (aqueous inorganic acids at RT) | 3 | 2 | 3 | 2 | |
| Alkalis (aqueous inorganic alkalis at RT) | 3 | 2 | 3 | 2 | |
| Water (at 100 °C, distilled) | 3 | 2 | 3 | 2 | |
| Weather and ozone | 1 | 1 | 3 | 1 | |
Table 1: Comparison of Properties of IIR with Other Rubber and Silicone Materials
Cost comparison between IIR and other elastomers
In terms of price, IIR is in the mid-range compared to alternative elastomers. IIR is more expensive than NR, SBR, EPDM, and NBR (factor x1.0 each) and is also above IR (factor x1.1). Compared to higher-priced materials like VMQ (factor x1.6), IIR is at a similar level, but remains significantly cheaper than FKM (factor x3.7).
| Mate rial | NR | SBR | EPDM | NBR | IR | IIR | VMQ | FKM |
|---|---|---|---|---|---|---|---|---|
| Cost factor | x 1,0 | x 1,0 | x 1,0 | x 1,0 | x 1,1 | x 1,6 | x 1,6 | x 3,7 |
Table 2: Cost comparison of IIR with other rubber and silicone materials
