Chloroprene (CR): highly resistant to weathering, ozone and ageing with good mechanical strength
1. what is CR?
CR (chloroprene rubber) is a synthetic, polarised elastomer based on polymerised 2-chloro-1,3-butadiene and is chemically known as polychloroprene.
Thanks to its chlorinated polymer structure, CR has good resistance to ozone, weathering, ageing and many oils and chemicals. The typical application temperature range is between approx.
-40 °C and +100 °C.
CR combines solid mechanical strength, good abrasion resistance, and moderate oil resistance with relatively low flammability. Compared to NBR, CR offers better weathering and ozone resistance but lower resistance to aliphatic fuels. Compared to EPDM, CR has higher oil resistance but does not achieve EPDM's resistance to hot water and steam.
CR is therefore frequently used in the manufacture of Rubber-metal bushes, where weather resistance and useful oil/chemical resistance are required, without resorting to expensive special materials.
How was chloroprene created?
CR is one of the oldest industrially produced synthetic rubbers. It was developed in the early 1930s as part of the search for alternative elastomer materials to natural rubber, which was strategically critical at the time. With the successful polymerisation of chloroprene, the industrial production of an elastomer with significantly improved resistance to ageing and weathering was achieved for the first time, making CR rubber highly relevant from an early stage.
What is chloroprene and what is neoprene?
CR is the technical term for polychloroprene in accordance with international standards, while neoprene was originally a trade name for chloroprene rubber. The term neoprene was coined in the 1930s for the first commercial CR qualities and has since established itself as a colloquial term.
In today's technical usage, CR stands for the material group, while neoprene is mostly used informally or for marketing purposes.
What is the chemical structure of CR?
The chemical basis of chloroprene rubber is the monomer 2-chloro-1,3-butadiene (C₄H₅Cl), more commonly known as chloroprene. Chloroprene is a colourless liquid at room temperature. Emulsion polymerisation of this monomer produces polychloroprene, which is designated CR. The polymer structure consists of an unsaturated carbon backbone chain, to which chlorine atoms are bonded at irregular intervals. These chlorine atoms are crucial as they increase the material's polarity, thereby significantly contributing to the special properties of CR.
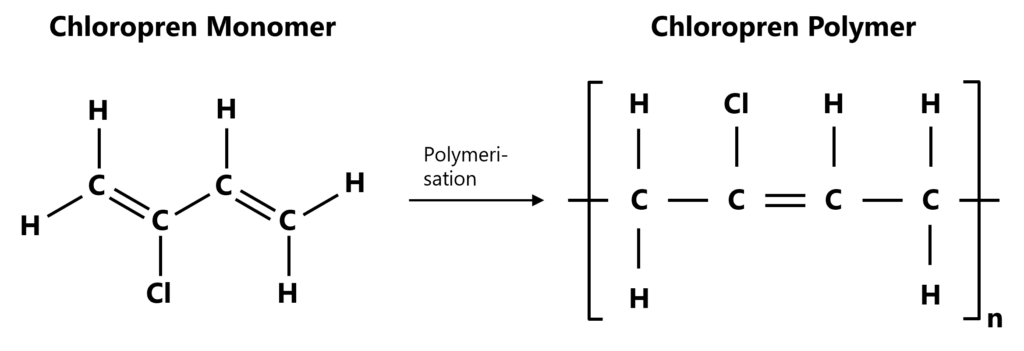
Figure 1: Chemical process from a chloroprene monomer (left) through polymerisation to a chloroprene polymer (right)
Während Naturkautschuk (NR) vollständig aus Isopren-Einheiten besteht, unterscheidet sich CR durch die eingebaute Chlorgruppe. Ein charakteristisches Merkmal von CR ist seine teilweise kristalline Struktur. Obwohl das Polymer überwiegend amorph vorliegt, können unter bestimmten Bedingungen kristalline Bereiche entstehen, die für eine verbesserte mechanische Festigkeit sorgen. Diese Mikrostruktur erklärt, warum Polychloropren in der Praxis eine gute Balance zwischen Elastizität und Festigkeit aufweist. Im Gegensatz zu unpolaren Synthesekautschuken wie SBR oder BR weist CR durch die Chlor-Substitution eine reduzierte Reaktivität gegenüber Schwefel auf. Das bedeutet, dass klassische Schwefelnetzwerke weniger stabil wären.
2. CR properties
Chloroprene rubber is characterised by a unique combination of mechanical, chemical and physical properties that set it apart from other synthetic rubbers. These properties are primarily based on the chlorine substitution in the polymer structure and the special vulcanisation chemistry.
Standards & guidelines from CR
CR is categorised as chloroprene rubber in accordance with international classification standards ISO 1629 and ASTM D1418. These standards are used to categorise materials, but do not specify any specific property values.
Mechanical parameters are tested on the basis of general elastomer testing standards, including ISO 37 (tensile test), ISO 48 (hardness), ISO 34 (tear propagation resistance) and ISO 815 (compression set). Thermal ageing resistance is usually assessed using heat ageing tests in accordance with ISO 188.
Tests in accordance with ISO 1817 are used to assess media resistance, in which changes in volume, mass and properties are determined after defined media contact.
Purity & cleanliness requirements
In addition to mechanical and chemical characteristics, application-specific purity and cleanliness requirements can be relevant for CR compounds. These are not defined by independent standards, but result from process- or customer-specific requirements.
The focus is on extractable components, migratable additives and particulate impurities. The actual purity is determined by the compound composition, the raw material selection and the process control. In the case of CR, purity requirements do not represent a general material parameter, but rather the result of a targeted material design.
Limits of chloroprene
Despite its balanced property profile, chloroprene (CR) has typical material limitations. Its resistance to mineral oils, fuels and aromatic hydrocarbons is limited and is significantly lower than that of NBR or FKM. The continuous thermal service temperature is also limited compared to high-performance elastomers.
Permanent thermal stress can lead to accelerated ageing, especially if the cross-linking or formulation design is unfavourable. In addition, the elasticity and rebound resilience is lower than that of natural rubber or highly elastic synthetic rubbers.
3. chloroprene production & processing
CR rubber is processed using the usual production methods for elastomers. Shaping is mainly carried out by compression moulding, transfer moulding or injection moulding, depending on the component geometry, quantity and required dimensional accuracy. CR is also extruded, for example for profiles, hoses or semi-finished products, which are then customised or further processed.
CR rubber is relatively easy to process, but requires a controlled manufacturing process as the material tends to release hydrogen chloride during processing. Uncontrolled release of hydrogen chloride during processing leads to damage to the polymer structure, as a result of which the resulting component may exhibit inhomogeneous mechanical properties, reduced ageing resistance and limited long-term stability.
Vulcanisation & cross-linking
The elastic properties of CR rubber are produced by vulcanisation, which creates a three-dimensional polymer network. Due to the chlorine-containing polymer structure, the cross-linking chemistry of CR differs significantly from that of non-polar elastomers. This results in two fundamentally possible cross-linking systems, which differ in their technical significance and the resulting property profile.
Metal oxide cross-linking
Metal oxide vulcanisation is the technically preferred and industrially established crosslinking process for CR rubber. Zinc oxide and magnesium oxide are used as crosslinking agents. These metal oxides have a dual function: on the one hand, they bind the hydrogen chloride released during processing and vulcanisation, thereby preventing structural degradation of the polymer. Secondly, they enable the formation of stable cross-linking structures within the polymer network.
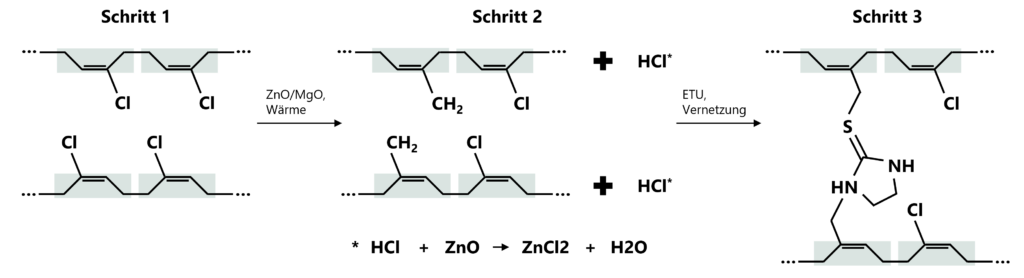
Figure 2: Crosslinking of chloroprene: In step 1 (left), uncrosslinked polychloroprene chains are present. In step 2, chemical cross-linking begins. In step 3 (right), a three-dimensionally cross-linked elastomer network is formed.
The resulting elastomer is characterised by high thermal stability, very good resistance to ageing and ozone and a balanced mechanical property profile.
Sulphur crosslinking
During sulphur crosslinking, the polymer reacts via the remaining double bonds in a similar way to natural rubber or styrene-butadiene rubber. Elastic polysulphide bridges are formed, which give the material a high resilience and good dynamic properties. However, the chlorine atom deactivates the double bond against typical sulphur radicals. As a result, sulphur networks form more slowly, are less stable and are often less reproducible than with non-polar rubbers. Sulphur vulcanisation is therefore rarely if ever used.
4. material comparison: CR vs. other elastomers
For CR, comparisons with NBR, NR and EPDM are particularly meaningful, as these materials are often discussed as alternatives in material selection.
| Chloroprene | Nitrile Rubber |
Natural rubber | Ethylene- Propylene diene Rubber |
||
| International abbreviation | CR | NBR | NR | EPDM | |
| Hardness range (in Shore) | 20A – 90A | 20A – 75D | 25A – 70D | 20A – 95A | |
| Mechanical Properties for Room temperature |
Tear resistance | 3 | 3 | 4 | 3 |
| Elongation at break | 3 | 3 | 3 | 3 | |
| Rebound resilience | 2 | 2 | 4 | 3 | |
| Tear propagation resistance | 3 | 2 | 4 | 3 | |
| Abrasion resistance | 3 | 2 | 3 | 3 | |
| Compression set | at max. continuous operating temperature | 2 | 1 | 1 | 0 |
| at room temperature | 0 | 0 | 0 | 0 | |
| Thermal behaviour | Cooling behaviour (Tg) up to °C | -40 | -30 | -55 | -50 |
| Max. Continuous operating temperature up to °C | 100 | 110 | 70 | 130 | |
| Resistance to | Petrol | 2 | 2 | 1 | 1 |
| Mineral oil (at 100 °C) | 2 | 3 | 1 | 1 | |
| Acids (aqueous inorganic acids at RT) | 2 | 2 | 2 | 3 | |
| Alkalis (aqueous inorganic alkalis at RT) | 2 | 2 | 2 | 3 | |
| Water (at 100 °C, distilled) | 2 | 2 | 2 | 3 | |
| Weather and ozone | 2 | 2 | 1 | 3 | |
Table 1: Material comparison of chloroprene (CR) with nitrile butadiene rubber (NBR), natural rubber (NR) and ethylene propylene diene rubber (EPDM)
What does CR cost?
Compared to other elastomers, CR has a medium cost level and is positioned between standard and high-performance materials.
| Material | EPDM | NBR | CR | TPE/TPU | Silicone (LSR) | Silicone (HTV) | HNBR | FKM |
|---|---|---|---|---|---|---|---|---|
| Cost factor | x 1,0 | x 1,0 | x 1,2 | x 1,3 | x 1,4 | x 1,8 | x 2,9 | x 3,7 |
Table 2: Cost comparison of chloroprene (CR) with other common elastomers
What distinguishes CR from NR?
Compared to natural rubber, CR has significantly better resistance to ageing, ozone and weathering. Natural rubber, on the other hand, offers greater elasticity and rebound resilience, but is much more sensitive to environmental influences.
What distinguishes CR from EPDM?
CR has a higher resistance to oils and greases than EPDM, while EPDM has superior resistance to water, vapour and weathering. CR is therefore positioned between oil-resistant and weather-resistant elastomers with a balanced property profile.
What distinguishes CR from NBR?
NBR shows significantly better resistance to oils and fuels, while CR is more resistant to ozone, ageing and flames. CR is therefore used where environmental resistance is more important than maximum oil resistance.
Mehr zum Material CR und zur Produktion von Gummiform- und Verbundteilen finden Sie in unserem ausführlichen Gummi-Lexikon, dem Glossary.
