BR (butadiene rubber) - abrasion-resistant and dynamically resilient for durable moulded rubber parts
1. introduction & history of butadiene rubber
BR (butadiene rubber) is a synthetic, unsaturated elastomer based on polymerised 1,3-butadiene and is chemically referred to as polybutadiene.
BR is characterised by very high abrasion resistance, excellent elastic recovery, low hysteresis losses and very good low-temperature flexibility. The typical application temperature is between around -90 °C and +100 °C.
The material is primarily used in tyre treads, technical moulded rubber parts, damping elements and dynamically stressed components. Compared to natural rubber (NR), BR offers better low-temperature flexibility and lower heat generation under dynamic stress, but does not achieve the same tear resistance. Compared to SBR, BR has higher elasticity and lower internal damping, but also has only limited resistance to ozone, weathering and mineral oils.
In Deutschland entwickelte die IG Farben Ende der 1920er Jahre den unter dem Handelsnamen Buna (Butadien-Natrium) vertriebenen Synthesekautschuk, der unter Zuhilfenahme von Natriumkatalysatoren polymerisiert wurde. Derivate waren Buna-BR (entspricht heute BR), Buna-S (entspricht heute SBR) und Buna-N (entspricht heute NBR).
The decisive further development came in the 1950s with the use of new catalyst systems such as Ziegler-Natta or neodymium catalysts. This made it possible to control the polymer structure more precisely, enabling the targeted synthesis of cis-1,4-rich polybutadiene types. These have a regular and flexible chain structure that provides the desired properties of high low-temperature flexibility, high rebound resilience, low heat generation under dynamic load and good resistance to ageing and abrasion.
Today, the starting material 1,3-butadiene is mainly obtained by steam cracking naphtha (crude petrol) and polymerised in highly specialised plants. At the same time, efforts are being made to synthesise butadiene from bioethanol as a sustainable alternative.
2. chemical composition of BR
Butadiene rubber (BR) is produced from the polymerisation of the gaseous 1,3-butadiene monomer. Chemically, it is an unsaturated polymer, as double bonds are still present in the main chain after polymerisation. These double bonds are crucial for subsequent vulcanisation, as they serve as reaction sites for sulphur or peroxide cross-linking.
The repeating unit consists of four carbon atoms with a double bond. If the hydrogen atoms on the double bond are on the same side, this is referred to as a cis configuration. If the hydrogens on the double bond are opposite each other, this is called a trans configuration. Such isomers (same structural formula, different arrangement) have fundamentally different chemical and physical properties.
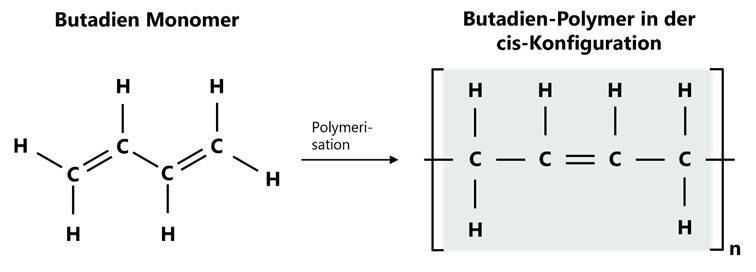
Figure 1: Polymer structure of polybutadiene (BR).
In industrially produced high-performance BR grades, a cis-1,4 proportion of over 95 % is usually aimed for compared to trans-1,4 and 1,2 units. A polymer in cis configuration is particularly flexible due to the more strongly angled chain structure and has a very low glass transition temperature (Tg ≈ -100 °C), which improves low-temperature resistance and elasticity. In contrast, trans chains are more elongated and tend to become brittle at low temperatures due to crystallisation.
Vulcanisation mechanisms of butadiene rubber
A key feature of BR is the unsaturated main chain, which is very reactive to oxygen and ozone due to the double bonds. Without suitable protection, for example by antioxidants, BR tends to age and become brittle. At the same time, this unsaturated structure enables the use of various vulcanisation systems, including sulphur, peroxide and resin cross-linking. In industrial practice, sulphur oxidation dominates, as it provides a particularly favourable combination of elasticity, abrasion resistance and thermal stability of the cross-linked polymer.
During sulphur crosslinking, sulphur chains of up to eight atoms in length bind to the BR, linking the polymer chain to form a three-dimensional network. The double bonds facilitate cross-linking, but remain in place even after vulcanisation. The length of the resulting sulphur bridges significantly determines the properties of the material: long polysulphide bridges lead to high elasticity, while short di- and monosulphide bridges result in greater thermal stability and hardness. Only a small proportion of the crosslinkable sites - around 1-3% - are actually utilised. A stronger conversion would make the material too hard and brittle.
Polysulphide sulphur chains can migrate along the polymer chains, which favours the reduction of local stresses and certain self-healing effects. On the one hand, this increases the tear propagation resistance, but also the compression set. These two qualitatively opposing mechanisms are inevitably coupled with each other. In practice, BR is often not used as a single rubber, but in blends with other synthetic or natural rubbers such as NBR, SBR or NR in order to achieve an optimum property profile - for example, the high abrasion resistance of BR combined with the better ageing resistance and wet adhesion of SBR and NR. Better resistance to oil and fuels is achieved by mixing with NBR.
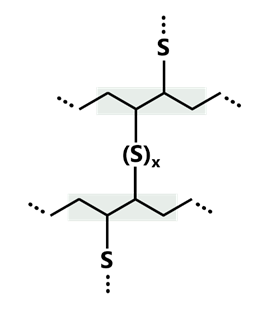
Figure 2: Schematic representation of cross-linking by sulphur bridges of BR.
3. properties & special features of BR
Butadiene rubber has high rebound resilience combined with low hysteresis losses. The uniform chain structure enables efficient energy transfer under dynamic stress, which means that less energy is lost in the form of heat. This results in a material with high fatigue strength and excellent mechanical resilience. In addition, BR has above-average abrasion resistance, but is only suitable to a limited extent or not at all for applications where weather or oil/fuel resistance is important.
Due to its low glass transition temperature, BR remains elastic even at very low temperatures and retains its resilience, which makes it ideal for applications where low-temperature flexibility is crucial (down to approx. -70 °C).
| International abbreviation | BR | NR | SBR | NBR | |
| Hardness range (in Shore) | 40A – 90A | 25A – 70D | 20A – 70D | 20A – 75D | |
| Mechanical Properties for Room temp. |
Tear resistance | 2 | 4 | 3 | 3 |
| Elongation at break | 3 | 3 | 3 | 3 | |
| Rebound resilience | 4 | 4 | 3 | 2 | |
| Tear propagation resistance | 1 | 4 | 3 | 2 | |
| Abrasion resistance | 4 | 3 | 3 | 2 | |
| Compression set | at max. continuous operating temperature | 2 | 1 | 1 | 1 |
| at room temperature | 1 | 0 | 0 | 0 | |
| Thermal behaviour | Cooling behaviour (Tg) up to °C | -90 | -55 | -45 | -30 |
| Max. Continuous operating temperature up to °C | 100 | 70 | 90 | 110 | |
| Resistance to | Petrol | 1 | 1 | 1 | 2 |
| Mineral oil (at 100 °C) | 1 | 1 | 1 | 3 | |
| Acids (aqueous inorganic acids at RT) | 2 | 2 | 2 | 2 | |
| Alkalis (aqueous inorganic alkalis at RT) | 2 | 2 | 2 | 2 | |
| Water (at 100 °C, distilled) | 2 | 2 | 2 | 2 | |
| Weather and ozone | 2 | 1 | 1 | 2 | |
Table 1: Properties of selected materials: 0 = unsuitable, 8 = very suitable
Compared to other common rubbers such as natural rubber (NR), styrene-butadiene rubber (SBR) or acrylonitrile-butadiene rubber (NBR), BR rubber is primarily used in highly dynamic applications. Compared to NR, BR is less susceptible to heat cracking. SBR achieves slightly better dimensional stability at higher temperatures, while BR scores with better low-temperature flexibility and lower hysteresis losses. Although NBR is significantly more resistant to oil and fuel, it does not come close to the cold elasticity or wear resistance of BR.
4. areas of application of BR
The fields of application for butadiene rubber (BR) result from its combination of low-temperature flexibility, low hysteresis, high rebound resilience and very good abrasion resistance. These properties make it a favoured material for components that are subject to continuous dynamic loads and at the same time require high energy efficiency.
In the tyre industry, butadiene rubber is a central element of modern tread compounds. It is usually combined with natural rubber (NR) or styrene-butadiene rubber (SBR) in order to achieve an optimum ratio of abrasion resistance, wet grip and tear resistance. Typical compounds contain 20-40 phr* BR. For special applications such as high-performance or winter tyres, however, the BR content can be significantly higher, as its low-temperature properties and low rolling resistance are particularly valued here.
In conveyor and drive technology, butadiene rubber is usually used as a mixture with NR or NBR. NR increases tear resistance and processing properties, while NBR provides additional oil and fuel resistance. This results in belts, roller coverings or conveyor belts with a long service life and good resistance to mechanical wear.
In the footwear and sporting goods industry, BR is used both in blends and in almost pure form when maximum abrasion resistance and elastic shock absorption are required. Soles and sports surfaces in particular benefit from the wear resistance and elasticity of the material.
In special applications such as vibration elements, seals or shock-absorbing components, the BR content is varied according to requirements in order to achieve an optimum compromise between flexibility, wear resistance and durability.
*phr: “parts per hundred rubber” Reference value is the total mass of all synthetic and natural rubbers used, excluding fillers, plasticisers, etc.
Mehr zum Material BR und zur Produktion von Gummiform- und Verbundteilen finden Sie in unserem ausführlichen Gummi-Lexikon, dem Glossary.
